The Metamorphic Petrology of Ice Cream
I was struck by the similarity of these two images. Which one does your dentist want you to eat?
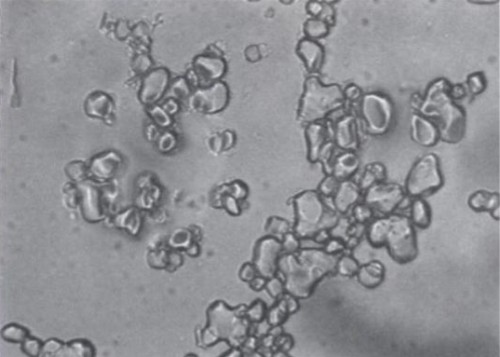
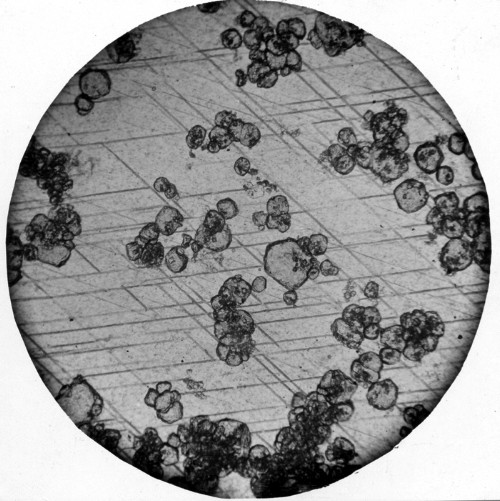
 I won’t speculate about anyone’s dentist’s motivations, but the top image comes from The Science of Ice Cream, and the bottom from the USGS. I’m not sure what conditions the ice cream was stored under, but the USGS image is from a limestone that had been stored next to a piping hot intrusion of monzonite, and partially baked into marble.
I won’t speculate about anyone’s dentist’s motivations, but the top image comes from The Science of Ice Cream, and the bottom from the USGS. I’m not sure what conditions the ice cream was stored under, but the USGS image is from a limestone that had been stored next to a piping hot intrusion of monzonite, and partially baked into marble.
Frozen desserts left in the freezer too long will undergo a similar process. The edges of each ice crystal are constantly exchanging water with the sugar solution. Small crystals, which have a lot of surface area, will tend to shrink especially fast while the more energetically favorable crystals (in the ice cream’s case, it’s just big ice crystals that are better than small ones, but in rocks it is usually a different mineral phase) will tend to grow. This process is accelerated at higher temperatures, which is why it’s a bad idea to leave a carton of ice cream sitting out on the counter even if it doesn’t obviously melt.
Here, the top image is of fresh ice cream, and the bottom is of the same ice cream after it’s sat in the freezer for 7 weeks:

Donhowe et al., 2020. Determination of ice crystal size distribution in frozen desserts. J. Dairy Sci. 74 (10).
It’s not freezer burn and off flavors from sitting next to fish fillets that make old ice cream taste bad – or it’s not just freezer burn and fish, anyway. Our perception of taste is closely linked to texture and mouthfeel, and those giant ice crystals are just yucky.
greg laden wrote:
This is brilliant. You must turn this into a lab for Geo 2020
Posted 21 Aug 2020 at 10:12 am ¶
Dr. Kate wrote:
Well, there’s another book going on my “must-read” list! (Why is it that all the books I want to write have already been written? Imagine the research for “The Science of Ice Cream!”)
Now that you explain it, it does kind of make sense that ice cream and parbaked marble might have similar structures. But it’s definitely not a comparison I would ever have thought to make.
Posted 21 Aug 2020 at 12:13 pm ¶
Bill wrote:
So, I’m guessing that in the case of the image of the partially formed marble the granules are calcium carbonate that has yet to undergo diagenesis and the lattice streaked area is the newly formed marble?
Posted 21 Aug 2020 at 4:25 pm ¶
Kim wrote:
To Bill:
The granules are teeny-tiny garnet crystals. (Green garnet, according to the caption on the USGS link that Maria gave. Not pure calcium carbonate in the starting material.)
It would be a bummer to bite into ice cream and hit a bite of garnet. Ouch.
Also, the possibility that my ice cream might metamorphose is a good excuse to eat it.
Posted 21 Aug 2020 at 4:52 pm ¶
Silver Fox wrote:
It’s apparent that if we took a vote on igneous v. metamorphic ice cream, that igneous would win hands down – whereas some of my favorite rocks are metamorphic!
Posted 21 Aug 2020 at 6:55 pm ¶
Bill wrote:
Thanks, Kim. I’ve afraid I have a dial-up connection that is at the moment, even for dial-up, dead slow, so I thought I’d just ask.
Posted 21 Aug 2020 at 7:29 pm ¶
Lab Lemming wrote:
So are we doing ice cream stratigraphy tomorrow?
Posted 21 Aug 2020 at 8:09 pm ¶
Kim wrote:
I suspect that sedimentary deposit of ice cream might involve a lot of hysterical crying from the five-year-old.
Posted 21 Aug 2020 at 9:13 pm ¶
Ibod Catooga wrote:
You’ll be pleased to know I just pooped my pants.
Thanks.
Posted 28 Aug 2020 at 1:43 am ¶
sefer ceka seferi wrote:
my name is sefer ceka seferi im a study of geography my from macedonia i live kicevo .
i love petrology, mineralorgy
i dont no very good spoke english
bye ceka
Posted 18 Feb 2020 at 1:50 pm ¶
Michael Dennis Stagg wrote:
If half of you understood petrology and your other half understood ice you might not make such blithering mistakes as has been pointed out by the garnet observers, the similarity just does not exist at all, which is probably why somebody wrote the baby is crying. No wonder some confused people have global warming and some write daft books on mineralogy and petrology with isotopes without understanding structures. But then I am not an isolated laboratory chemist and I do not choose to live on and exploit a subsiding coral Atoll in a volcanic region of plate tectonic boundary shifts as Australia moves ever into the deep blue wet thing Pacific … Muppets and then appear on TV complaining to the BBC their crops are damp.
Posted 02 Oct 2020 at 7:54 am ¶
kids dentist wrote:
As a dentist, I would prefer to eat the ice cream on the 2nd image.
Posted 11 Jan 2020 at 6:20 pm ¶